
May 13, 2022 | Mike Kim
Testing is usually perceived as the activity performed AFTER development is complete. If you’re familiar with the medical device industry, you probably heard about Verification & Validation testing: “was the product built right?” or “was the right product was built?” While Verification and Validation is incredibly important, I do want to share a recent example of how testing has helped with our development.
We have a new device in development that requires the skin to be wiped with an alcohol wipe (70% isopropanol). The wipe is part of the device administration protocol and is needed for our device to function properly as it prepares the skin by removing dirt, soap, cosmetics and other debris that may impact the device’s functionality.
A health care professional helping to test our device recently provided feedback on encountering brown residue on a patient’s wrist which was identified as Betadine (Povidone-Iodine). Betadine is an antiseptic used to disinfect the skin however it also stains the skin, and at the time, we didn’t know how our device would perform when encountering this situation.
We are fortunate to have an Optical Scientist on our core project team who was quick to point out that iodine has certain characteristics that will affect how our device functions. With his input and a quick trip to the local pharmacy, we quickly devised and executed a couple ad-hoc tests to help understand how Betadine impacts the device.
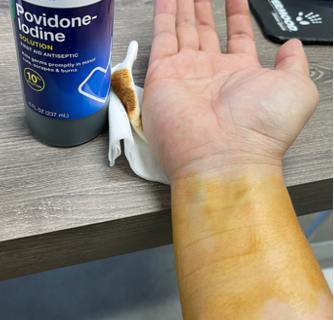
First was a simple visual test to see if Betadine and the staining of the skin can be fully removed with our current protocol with using an alcohol wipe. It was observed that using just a single wipe was not sufficient in removing the stain however we did confirm that multiple wipes did successfully remove all visual remnants of Betadine from the skin.
Additional tests were performed to fully understand what the non-removal, partial-removal, and full-removal of Betadine with alcohol wipes has with the device. After reviewing the data with our Optical Scientist and our core project team, we’ve addressed this situation by updating risk files, device administration protocol, and training materials.
Although, it is impossible to identify all risk scenarios for a new device especially during development, testing during the development phase can help provide valuable feedback especially if it’s from real health care professionals in real environments.